Role of HyTest in Cardiac Troponin Detection and Myocardial Infarction Diagnosis
At HyTest, our specialization in cardiac troponin (cTn) detection spans nearly three decades. The journey began in 1994 with the introduction of the first anti-cardiac troponin I (cTnI) monoclonal antibodies (MAbs) to the market. Over the years, we have generated and analysed thousands of cTn-specific antibodies. The unwavering dedication of our scientists to the intricate processes of developing, producing, and purifying this protein in its various forms has made it a cornerstone of our expertise.
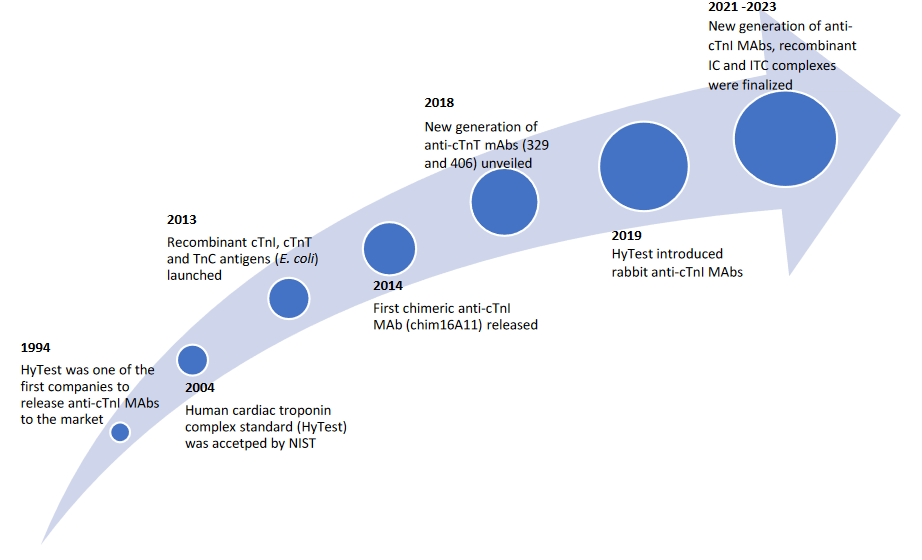
Highlights in the history of HyTest’s of Cardiac Troponins Development
Troponin complex plays an important role in the regulation of skeletal and cardiac muscle contraction. This protein consists of three subunits: TnT, TnI, and TnC. Specific isoforms of cTnT and cTnI, are unique to the myocardium. During myocardial infarction (MI), cardiac isoforms of troponin are released from damaged myocytes. These proteins have become guideline-recommended biomarkers, essential for diagnosing acute MI.
Development Highlights of Cardiac Troponin Antigens
In a 1997 study, HyTest scientists demonstrated for the first time that the cTnI molecule is not released in a free form as previously thought but is complexed with TnC. We suggested using antibodies for assay development that are not affected by the formation of the cTnI-TnC complex and could recognize both free and complexed cTnI forms.(1) Further research in 1999 involved comparing results from different cTnI assays by measuring cTnI concentration in clinical samples, we concluded that using the native troponin complex as the calibrator could reduce assay-to-assay variation.(2) Based on these findings, HyTest developed its native Cardiac Troponin I-C binary complex and I-T-C complex antigens. These have shown improved results when used as calibrators in developing cTnI assay. HyTest’s Troponin I-T-C complex has been selected by the AACC cTnI Standardization Subcommittee for international reference material. The standard reference material (SRM® 2921) can be accessed at the National Institute of Standards and Technology (more details are available on www.nist.gov). Over the years, we have developed recombinant cardiac troponins I, T and С (Cat. # 8RT17, Cat. # 8RTT5, Cat. # 8RSC4 and Cat. # 8RKC3) as well as Cardiac Troponin I-C complex (Cat. # 8ICR3) and recombinant Cardiac Troponin I-T-C complex (Cat. # 8ITCR) antigens as substitutes for the native ones. In the 1998 and 2018 article by Katrukha A et al. and Katrukha I et al., respectively, the central region of cTnI was characterized as the most stable part of the molecule. Currently, most diagnostic systems employ antibodies with epitopes located in the central part of cTnI. (3,4) Building on the research, in 2023, we finalized the launch of the recombinant chimeric human Cardiac Troponin binary Complex (Cat. # 8IFC20). This unique complex features the most stable part of cTnI (fragment 28-110) linked with TnC by a 20-aar flexible linker. We invite you to delve deeper into our cTn antigen products by exploring our Troponin Booklet.
A Comprehensive Guide to Our Troponin Antibodies
Detecting cTn in patients' blood can be challenging, especially when it comes to cTnI. Quantitative measurement can be influenced by factors such as phosphorylation, proteolytic degradation, the cross-reactivity with skeletal TnI and autoantibody interference (Katrukha AG et al. 1999).(5) The influence of these factors on the interaction of antibodies with cTnI is multidimensional. Our growing comprehension, however, has identified key requirements for creating sensitive, quantitative immunoassays that enable accurate measurements of troponins in blood samples. From the initial offering of anti-cTnI MAbs, we have vastly expanded our portfolio. After almost three decades, cTnI antibodies have evolved through many generations. Our portfolio now includes over 30 specially selected anti-cTnI MAbs, specific to the various epitopes on the cTnI molecule. Building on our commitment to nonstop improvement, 2023 witness the emergence of new anti-cTnI “Y” generation antibodies, including Y303, Y306, Y303, Y101, Y302, Y501, Y502, Y504, Y505, Y601 and Y603 (Cat. # RC4T21). These MAbs are specially developed to target three regions of cTnI (approximately bordered by 20-40, 80-95 and 160-190 aar) that are crucial for the sensitive and specific diagnosis of acute MI.
We offer not only anti-cTnI antibodies but also antibodies specific to cTn complex. Inspired by studies conducted by HyTest scientists in the late 1990s, we developed several MAbs specifically targeting the cTn complex. Of these, 20C6cc stands out as a star product in HyTest, having gained widespread recognition and acceptance by customers around the world. These anti-cTn complex MAbs can be used in high-sensitivity cTnI sandwich immunoassay together with antibodies specific to human cTnI (Cat. # RC4T21) and TnC (Cat. # 4T27cc). Beyond conventional 1+1 format, we can also recommend a multi-MAb approach that enables the detection of cTnI in a 1+2 or 2+2 design.
And lastly, anti-cTnT MAbs 329cc and 406cc (Cat. # 4T19cc) certainly merit attention. In a 2023 article by Katrukha IA et al., our researchers bordered major cTnT fragment and quantified their relative abundance in the blood at different times after MI. cTnT was present in the blood of MI patients as 23 proteolytic fragments. The cTnT region approximately bordered by aar 69-158 is a promising target for antibodies used for measurement of total cTnT.(6) 329cc and 406cc have superior sensitivity (limit of detection better than 0.3 ng/L) and high specificity (no cross-reaction to cTnI or to skeletal isoforms of TnT up to 30 µg/L).
Beyond the products highlighted, we invite you to dive into our extensive range of antibody offerings. Discover a diverse array of options beyond troponin antibodies, all meticulously detailed in our Troponin Booklet.
Hytest’s Commitment to Cardiac Troponin Research
cTn presents unique challenges as a complex analyte. Nonetheless, our expanding R&D team at HyTest remains at the forefront, pushing the boundaries in research and product development. Our scientists regularly publish articles in peer-reviewed journals, highlighting our commitment to rigorous scientific inquiry (you can find these articles in the Resources section on the HyTest website). Additionally, our active participation in IFCC working groups for troponin I standardization further showcases our dedication to excellence. As we look into the future, our commitment to advancing the field of cardiac diagnostics remains strong. With each advancement, we stride towards a world where timely, accurate diagnoses become standard, aiming for improved outcomes for MI patients globally.
References:
1. Katrukha AG et al. Troponin I is
released in bloodstream of patients with acute myocardial infarction not in
free form but as complex. Clin. Chem. 1997; 43(8):1379-1385.
2. Katrukha AG et al. New approach
to standardization of human cardiac troponin I (cTnI). Scand. J. Clin. Lab. Invest.,
Suppl. 1999, 230:124-127.
3. Katrukha AG et al. Degradation of
cardiac troponin I: implication for reliable immunodetection. Clin. Chem. 1998;
44:12 2433–2440. doi: 10.1093/clinchem/44.12.2433.
4. Katrukha IA et al. Full-Size
Cardiac Troponin I and Its Proteolytic Fragments in Blood of Patients with
Acute Myocardial Infarction: Antibody Selection for Assay Development. Clin.
Chem. 2018;64(7):1104-1112. doi: 10.1373/clinchem.2017.286211.
5. Katrukha AG et al. Biochemical
Factors Influencing Measurement of Cardiac Troponin I in Serum. Clin Chem Lab
Med 1999; 37(11/12):1091–1095. doi: 10.1515/CCLM.1999.159.
6. Katrukha IA et al. Fragmentation of human cardiac troponin T after acute myocardial infarction. Clin. Chem. 2023; 542:117281. doi: 10.1016/j.cca.2023.117281.